 Notice how Na after in the second I.E, Mg in the third I.E., Al in the fourth I.E.\): Elements categorized into metals, non-metals and metalloids. These are the ionization energies for the period three elements. Therefore, it requires less energy to remove one of their valence electrons Table 1: Ionization Energies of certain elements (1st IE, 2nd IE, etc) Element 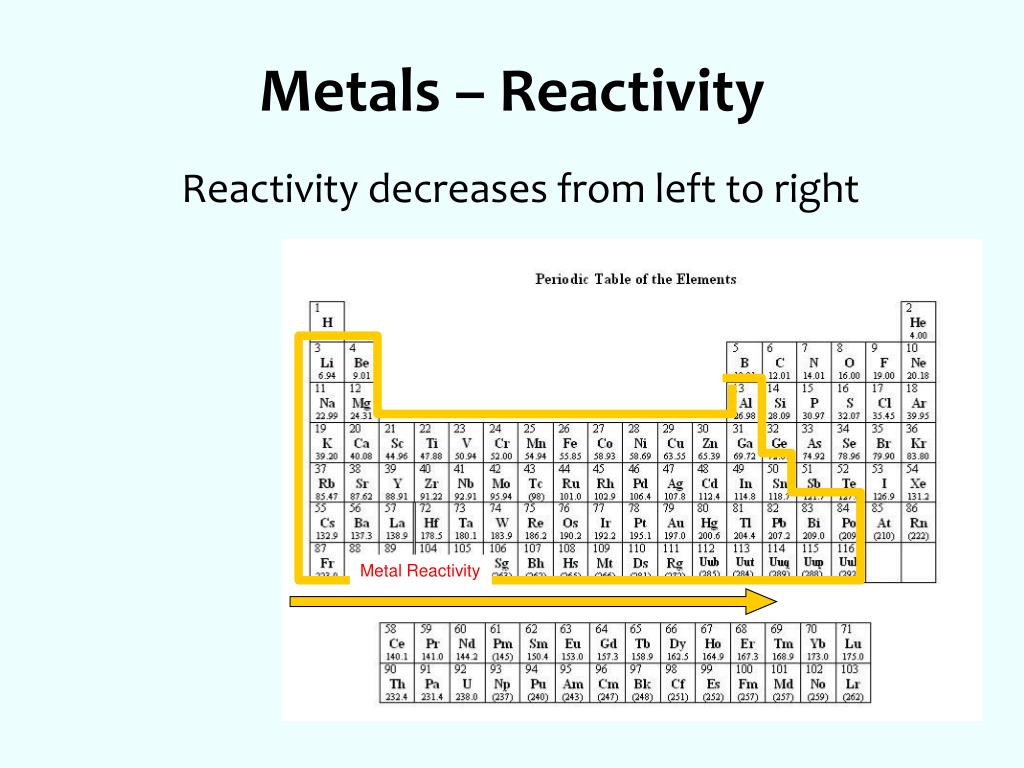
Additionally, elements in the left corner have a low ionization energy because losing an electron allows them to have the noble gas configuration. Patterns and trends in the periodic table Chemists observe patterns in different properties of elements as they are arranged in the periodic table. Reactivity: Reactivity describes the ability of a molecule or atom to undergo a chemical reaction, followed by a release in energy. When moving from left to right across the periodic table, we observe a decrease in the chemical reactivity of metals, whereas for. These trends are influenced by factors such as ionization energy and electronegativity. The highest amount of energy required occurs with the elements in the upper right hand corner. Chemical reactivity trends in the periodic table play a crucial role in understanding the behavior and interactions of elements. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. 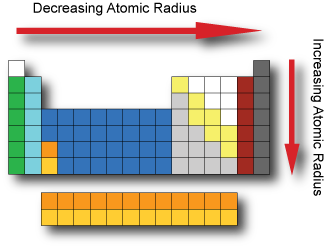
These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. The highest ionization energies are the noble gases because they all have high effective charge due to their octet formation and require a high amount of energy to destroy that stable configuration. The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. The specific arrangement of Group 1 elements on the periodic table allows us to make predictions about their reactivity. Ionization energies increase relative to high effective charge. Revise the history, patterns and structure of the periodic table including groups and periods group 1 metals transition metals non-metals reactivity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
